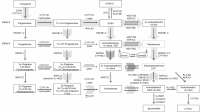
Adnrosterone is a downstream metabolite of DHT and just like DHT it is a 5-AR derived steroid. What is even more interesting is that androsterone is not just a metabolite of DHT but can be converted back to DHT as needed in target tissues where DHT is needed. In fact, androsterone has at least 2 different pathways through which is can convert back to DHT. See attached images for the various pathways through which androsterone converts back into DHT and 5-alpha androstanedione (another potent 5-AR androgen and also a precursor to DHT). Various studies have shown that androsterone sulfate is the only reliable biomarker of androgenic reserves. Levels of DHEA-S, while also well-correlated with androgenic activity, are also indicative of estrogenic reserves as DHEA can easily aromatize into estrone under the wrong circumstances. So, it is the levels of long-lived, non-aromatizable androgens like androsterone sulfate that are truly indicative of the androgenic status of the organism.
In vivo studies show that indeed, administration of androsterone elevates plasma levels of DHT and some other androgenic metabolites.
Androsterone - Wikipedia, the free encyclopedia
"...Androsterone, or 3α-hydroxy-5α-androstan-17-one, is an endogenous steroid hormone, neurosteroid, and putative pheromone.[1] It is a weak androgen with a potency that is approximately 1/7th that of testosterone.[2]In addition, it can be converted to dihydrotestosterone (DHT) from 3α-hydroxysteroid dehydrogenase and 17β-hydroxysteroid dehydrogenase, bypassing conventional intermediates such as androstenedione and testosterone, and as such, can be considered to be a metabolic intermediate in its own right.[3][4] Androsterone is also known to be an inhibitory androstane neurosteroid,[5][6] acting as a positive allosteric modulator of theGABAA receptor,[7] and possesses anticonvulsant effects.[8] The unnatural enantiomer of androsterone is more potent as a positive allosteric modulator of GABAA receptors and as an anticonvulsant than the natural form.[9] Androsterone's 3β-isomer is epiandrosterone, and its 5β-epimer is etiocholanolone."
The metabolism of 5alpha-dihydrotestosterone and androsterone by skin. - PubMed - NCBI
"...Androsterone was converted into the following metabolites : 5a-androstanedione, epiandrosterone, 5a-dihydrotestosterone, 5a-androstane-3a,l7/-?-diol and 5a-androstane-3&17/3-diol. 5a-Androstanedione was a major product in all incubations especially with rat preputial gland and rat skin. Epiandrosterone was a major product with the preputial gland. Rat skin did not convert androsterone into 5a-androstane-3p,17/?-diol."
Psychomotor and rewarding properties of the neurosteroids dehydroepiandrosterone sulphate and androsterone: effects on monoamine and steroid metabo... - PubMed - NCBI
"...DHEAS administration augmented concentrations of DHEA, pregnenolone, androstendiol and androstentriol in both brain structures, while androsterone injections increased brain levels of androsterone, epiandrosterone, 5α-dihydrotestosterone, and androstandiol. Present data document that although psychobehavioral and neurochemical effects of DHEAS and androsterone differ in several aspects; both neurosteroids have rewarding properties at certain dose ranges, suggesting their likely involvement in addictions, which entail different mechanisms."
In vivo studies show that indeed, administration of androsterone elevates plasma levels of DHT and some other androgenic metabolites.
Androsterone - Wikipedia, the free encyclopedia
"...Androsterone, or 3α-hydroxy-5α-androstan-17-one, is an endogenous steroid hormone, neurosteroid, and putative pheromone.[1] It is a weak androgen with a potency that is approximately 1/7th that of testosterone.[2]In addition, it can be converted to dihydrotestosterone (DHT) from 3α-hydroxysteroid dehydrogenase and 17β-hydroxysteroid dehydrogenase, bypassing conventional intermediates such as androstenedione and testosterone, and as such, can be considered to be a metabolic intermediate in its own right.[3][4] Androsterone is also known to be an inhibitory androstane neurosteroid,[5][6] acting as a positive allosteric modulator of theGABAA receptor,[7] and possesses anticonvulsant effects.[8] The unnatural enantiomer of androsterone is more potent as a positive allosteric modulator of GABAA receptors and as an anticonvulsant than the natural form.[9] Androsterone's 3β-isomer is epiandrosterone, and its 5β-epimer is etiocholanolone."
The metabolism of 5alpha-dihydrotestosterone and androsterone by skin. - PubMed - NCBI
"...Androsterone was converted into the following metabolites : 5a-androstanedione, epiandrosterone, 5a-dihydrotestosterone, 5a-androstane-3a,l7/-?-diol and 5a-androstane-3&17/3-diol. 5a-Androstanedione was a major product in all incubations especially with rat preputial gland and rat skin. Epiandrosterone was a major product with the preputial gland. Rat skin did not convert androsterone into 5a-androstane-3p,17/?-diol."
Psychomotor and rewarding properties of the neurosteroids dehydroepiandrosterone sulphate and androsterone: effects on monoamine and steroid metabo... - PubMed - NCBI
"...DHEAS administration augmented concentrations of DHEA, pregnenolone, androstendiol and androstentriol in both brain structures, while androsterone injections increased brain levels of androsterone, epiandrosterone, 5α-dihydrotestosterone, and androstandiol. Present data document that although psychobehavioral and neurochemical effects of DHEAS and androsterone differ in several aspects; both neurosteroids have rewarding properties at certain dose ranges, suggesting their likely involvement in addictions, which entail different mechanisms."
Attachments
Last edited: