noqcks
Member
- Joined
- Feb 8, 2020
- Messages
- 108
Hey everyone. I've seen the term "uncoupling" used around the forum. I'd like to share what I've learned about Mitochondrial Uncoupling. I also have a few questions.
From one of rays articles
I'm going to assume basic understand of mitochondria morphology and oxidative phosphorylation.
In the Electron Transport Chain (ETC), protons (H+) move from the mitochondrial matrix to the intermembrane space via the different ETC complexes (I, II, II, IV). The intermebrane space contains a higher concentration of H+ ions than the matrix — a concentration gradient is created, this is also sometimes referred to as the "proton-motive" force. There is an energy potential in the difference between these two concentrations that can be utilized. You can see all this happening in Figure 1.
Figure 1.
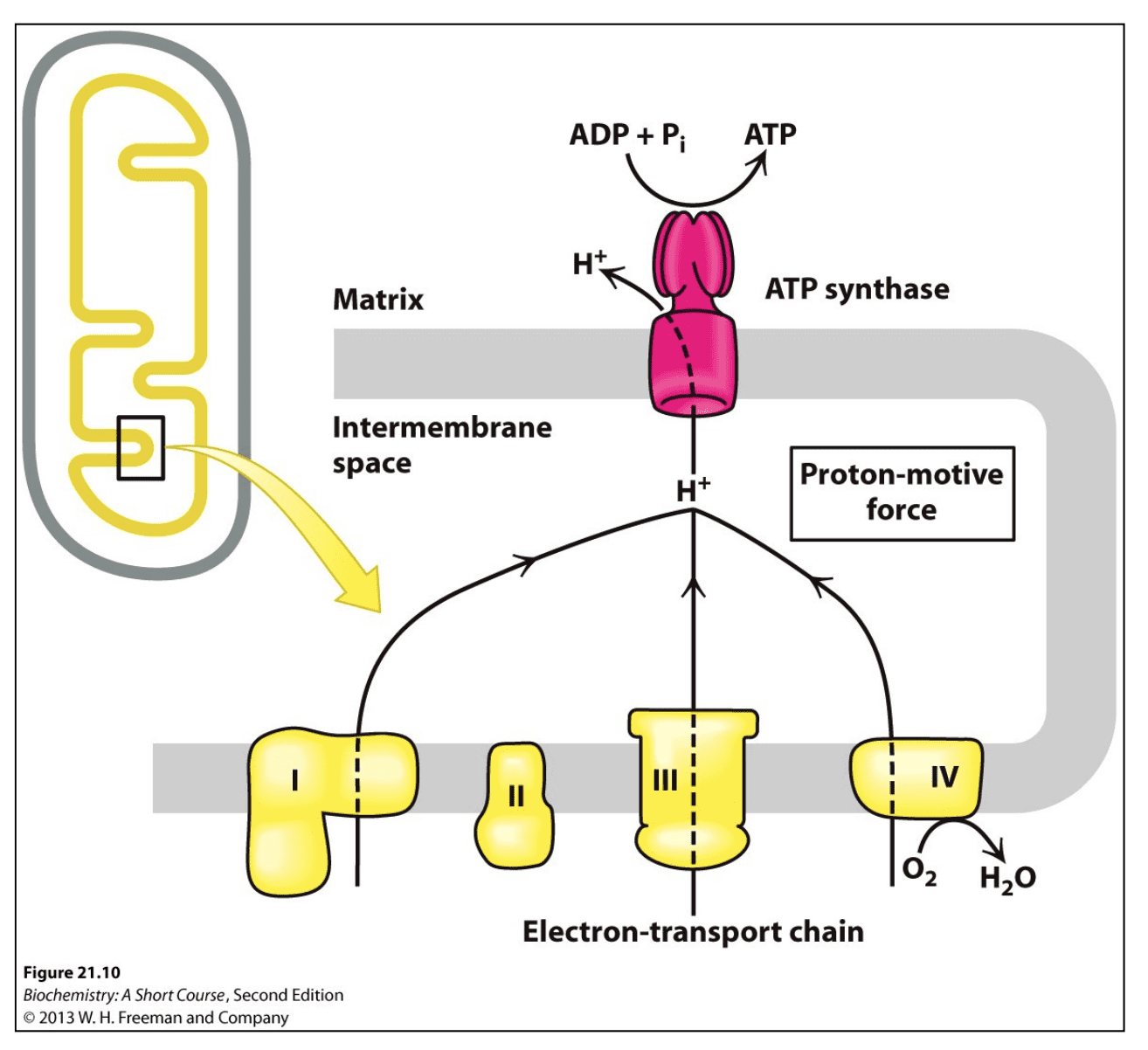
The H+ ions need to be moved back into the Matrix so that they can be re-used by the Electron Transport Chain. The H+ ions can move back into the matrix from the intermembrane space in one of two ways.

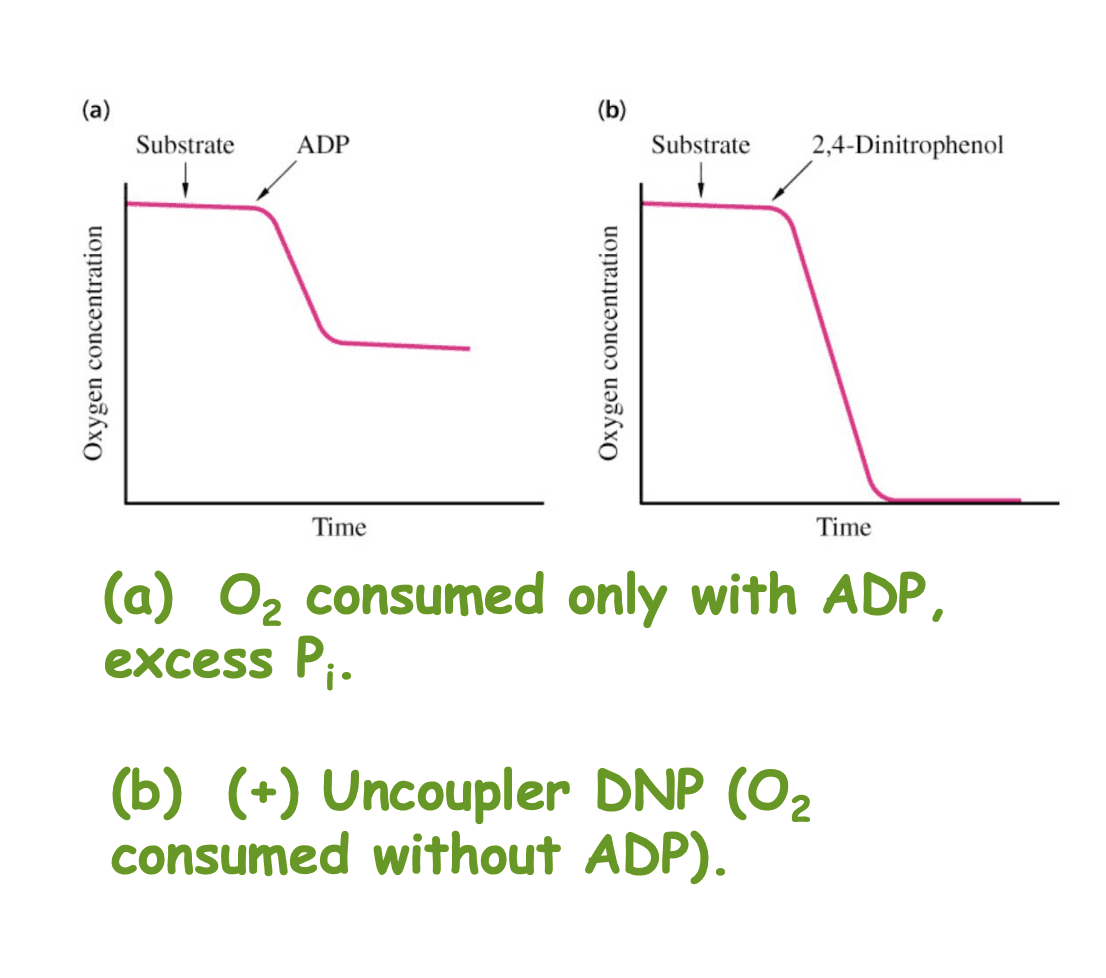
You can see in Figure 3, for example, what oxygen concentration looks like when an 2,4-Dinitophenol is used, an agent used for uncoupling oxidative phosphorylation.
Figure 3.
Some quotes from Peat
Questions I have for the smart people around here:
- How exactly does it reduce lactate production?
- Why is excessive uncoupling seen in cancer cells?
From one of rays articles
Uncoupling--In cellular respiration, oxidation of "fuel" in the mitochondrion is coupled to the phosphorylation of ADP, forming ATP. Uncouplers are chemicals that allow oxidation to proceed without producing the usual amount of ATP.
I'm going to assume basic understand of mitochondria morphology and oxidative phosphorylation.
In the Electron Transport Chain (ETC), protons (H+) move from the mitochondrial matrix to the intermembrane space via the different ETC complexes (I, II, II, IV). The intermebrane space contains a higher concentration of H+ ions than the matrix — a concentration gradient is created, this is also sometimes referred to as the "proton-motive" force. There is an energy potential in the difference between these two concentrations that can be utilized. You can see all this happening in Figure 1.
Figure 1.

The H+ ions need to be moved back into the Matrix so that they can be re-used by the Electron Transport Chain. The H+ ions can move back into the matrix from the intermembrane space in one of two ways.
- via ATP synthase. This enzyme uses ADP in the matrix and energy potential from the H+ gradient to produce ATP. Thus it is said that oxidative phosphorylation is coupled to the phosphorylation of ADP to produce ATP with ATP synthase. It is the limiting factor.
- via uncoupler proteins. Instead of ATPase, which needs ADP in the Matrix space to function, uncoupling proteins can be used to shuttle H+ across the membrane. This produces some heat in the process. I think higher body temps is what most people refer to when describing "uncoupling effects". It results in higher oxygen consumption, C02 production, lower lactate formation, and increases whole body energy expenditure. You can see the uncoupler protein in Figure 2. When using an uncoupler protein, respiration (oxidative phosphorylation) is said to be uncoupled.

You can see in Figure 3, for example, what oxygen concentration looks like when an 2,4-Dinitophenol is used, an agent used for uncoupling oxidative phosphorylation.
Figure 3.

Some quotes from Peat
Aspirin and thyroid (T3) increase uncoupling. A drug that used to be used for weight reduction, DNP, also uncouples mitochondrial metabolism, and, surprisingly, it has some of the beneficial effects of thyroid and aspirin. It stimulates the consumption of lactic acid and the formation of carbon dioxide.
The squirrel monkey, which on average weighs about 2 or 3 pounds as an adult, lives much longer than other mammals of its size, usually about 20 years, as long as 27. It has an extremely high rate of oxygen consumption. This is probably the result of natural uncoupling of the mitochondria, similar to that seen in long-lived mice. Mice with 17% higher resting oxygen consumption lived 36% longer than slow respiring mice of a related strain (Speakman, et al., 2004).
When mitochondria are “uncoupled,” they produce more carbon dioxide than normal, and the mitochondria produce fewer free radicals. Animals with uncoupled mitochondria live longer than animals with the ordinary, more efficient mitochondria, that produce more reactive oxidative fragments. One effect of the high rate of oxidation of the uncoupled mitochondria is that they can eliminate polyunsatured fatty acids that might otherwise be integrated into tissue structures, or function as inappropriate regulatory signals.
Questions I have for the smart people around here:
- How exactly does it reduce lactate production?
- Why is excessive uncoupling seen in cancer cells?
Last edited:
